|
Getting your Trinity Audio player ready...
|
The medical technology sector has garnered a lot of attention from investors in recent times and if you are looking into any such companies this morning that you could consider tracking Intelligent Bio Solutions Inc (NASDAQ:INBS).
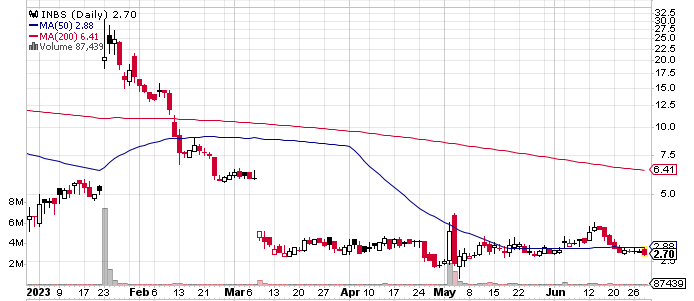
Following a fresh development the stock is trading up this morning and it could be the right time to get more of an idea about the actual announcement. The company revealed that it had been provided with guidance by the United States Food and Drug Administration with regard to the regulatory classification for its Intelligent Fingerprinting Drug Screening Cartridge.
In the past, the FDA had announced that the cartridge should be included in the 21 CFR 862.3650, Opiate Test System, which described a Class II device that would necessitate the submission of a pre-market notification 510 (k) and the clearance from the agency as well prior to the start of marketing.
The preliminary assessment made by the agency provided a clear pathway for Intelligent Bio Solutions as it started to work on the expansion of the product in the United States. It was also noted by the company that it was looking to submit its 510 (K) pre-market notification for its product.
The Chief Executive Officer and President of the company Harry Simeonidis spoke about the latest development as well. He noted that everyone at the company was pleased with the fact that it had received the preliminary assessment from the FDA for the Intelligent Fingerprinting Drug Screening Cartridge.
He went on to add that with the fast collection time and easier portability, the product had the potential of revolutionizing drug screening technology in the United States. The Intelligent Fingerprinting Drug Screening Cartridge is meant for detecting the presence of certain specific drugs in a person’s body through fingerprint sweat analysis.




